|
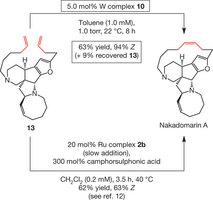
The total synthesis of nakadomarin A was realized through late-stage tungsten-catalysed ring-closing metathesis of the pentacyclic compound 13, and comparison with results delivered by Ruthenium catalysts.
[Credit: Nature DOI 10.1038/nature10563] |
Carbon–carbon double bonds, also referred to as alkenes or olefins, are present in many medicinally relevant and biologically active molecules. Hoveyda and MIT Professor Richard Schrock, a co-author of the Nature report, have collaborated since 1997 in developing new and effective catalysts for such transformations. Schrock shared the 2005 Nobel Prize in Chemistry for discovering one of the earliest types of olefin metathesis catalysts.
Alkenes exist as either the zigzag shaped trans olefin, or the E isomer, while others take the "U" shape of the cis olefin, or the higher-energy Z isomer. Z isomers require a catalyst that must be sufficiently active to be capable of promoting the chemical reaction while maintaining the cis olefin's U-shape geometry. Preserving both characteristics in a catalyst leads to reactions that deliver Z alkenes, which can be found in a large number of medicinally significant molecules and serve as starting materials for some of the most commonly used transformations in chemistry.
Ring-closing metathesis allows the formation of any ring size from readily available linear chains. However, the process is typically non-efficient or results in the formation of a significant amount of the undesired isomer, which often has less or no biological activity.
Using tungsten, the team found a catalyst that offers exceptional reactivity and selectivity while being sufficiently stable to air and moisture to make its use particularly attractive, said Hoveyda, who with Schrock co-founded the Swiss firm XiMo, which has licensed the new catalyst and its technology.
To demonstrate the utility of their catalysts, the team focused on epothilone C and nakadomarin A. Several other leading research groups in the world have previously prepared these biologically active natural products, but, in all cases, the critical ring closure step has been non-selective. This lack of selectivity badly damages the overall efficiency required to prepare these much sought after molecules, since ring formation comes at the tail end of a long sequence of reactions, sometimes as many as 20 steps.
"Losing half of your materials after you have spent so much time and energy and thought preparing it is nothing short of devastating, particularly when you cannot even separate the two isomers," Hoveyda says.
Epothilone C can be produced through fermentation, but creating various analogues by the same method would be extremely difficult. Nakadomarin A, found in marine environments, is only available in minute quantities. The highly selective process developed by the team will allow scientists to access significant quantities of these natural product as well as many of their analogs, which might prove to have more attractive therapeutic attributes.
The findings are the most recent from 14 years of collaboration between the Hoveyda and Schrock labs, work that has been supported by the National Institutes of Health since 1999. Also contributing to the study were Miao Yu and Chenbo Wang of Boston College and Andrew F. Kyle, Pavol Jakubec and Darren J. Dixon of the University of Oxford (UK).



