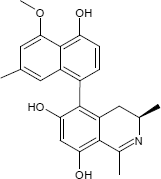
They are a substance class known for about 20 years and come from a liana from the African jungle.
A member of this Ancistrolikokine called natural product class is currently attracting attention: The variant E3 shown above is a promising drug candidate in the fight against certain forms of pancreatic carcinoma.

The IUPAC has updated the standard atomic weights of 14 chemical elements.
The atomic weights of aluminum, cobalt, gold, holmium, manganese, niobium, praseodymium, protactinium, rhodium, terbium, thulium and yttrium have been revised.

Usually, the movement of electrons in a real material is rather different from the flow of water in a river. However, in extraordinary materials like the metal oxide palladium cobaltate, "electron rivers" can exist, as predicted theoretically over fifty years ago and now demonstrated by scientists from the MPI CPfS [Image credit: MPI CPfS].

Mechanism for Radiation Damage Identified
Heidelberg scientists examine electronic decay processes with the aid of quantum chemistry.
What exactly are the processes when x-ray photons damage biomolecules with a metal centre?
[Credit: Internetchemie]
Last update: 08 November 2022
Perma link: https://www.internetchemistry.com
More chemistry: index | chemicals | lab equipment | job vacancies | sitemap
Internetchemistry: home | about | contact | imprint | privacy
© 1996 - 2023 Internetchemistry