Researchers at the RUB and from Berkeley have used metal complexes to modify peptide hormones.
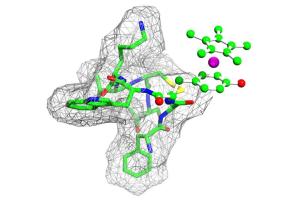
In the Journal of the American Chemical Society, they report for the first time on the three-dimensional structure of the resulting metal-peptide compounds [see below].
"With this work, we have laid the molecular foundation for the development of better medicines" says Prof. Raphael Stoll from the Faculty of Chemistry and Biochemistry at the Ruhr-University.
The team examined hormones that influence the sensation of pain and tumour growth.
Peptide hormones have many functions in the body
Hormones consisting of amino acids, the peptide hormones, convey bodily sensations such as pain and hunger, but also transmit growth signals. One example of this is insulin, which is important for the control of blood sugar levels. In interaction with specific receptors, the G-protein-coupled receptors, peptide hormones transport their messages to the cells. The hormones can be specifically chemically modified so that their effect changes, for example pain tolerance is lowered, or tumour growth inhibited. The German-Californian group of researchers has now found a new way to modify peptide hormones.
Metal complexes react with various peptide hormones
The first time they used a metal complex, namely, a rhodium compound, which reacts with the amino acid tyrosine. The precious metal rhodium is used as a catalyst in the synthesis of highly complex medicinal substances in the research laboratory as well as in industrial plants. Among other things, the researchers analysed the peptide hormone encephalin, which is important for the sensation of pain, and octreotide. The latter is a synthetic derivative of the growth hormone somatostatin, approved as a medicinal substance and already used in the treatment of certain tumours. The reaction with the metal complex was highly selective. Although the hormones consist of hundreds of atoms, the rhodium compound was always coordinated by the carbon ring of the tyrosine - the phenol ring.
Structure determined by NMR spectroscopy
The team also clarified the structure of the resulting metal-peptide complexes. "We hope to develop other metal-containing, peptide-like substances by building on these basic studies" says Prof. Dr. Nils-Metzler-Nolte of the Chair of Inorganic Chemistry I. "These could modulate the effect of naturally occurring peptide hormones and, for example, be used as a novel remedy for pain or cancer". For the project, the Californian colleagues made their knowledge of the special reactivity of the rhodium compound available. The researchers in Bochum contributed their experience with metal-peptides, the corresponding receptors and the structural analysis of biological macromolecules. "This again demonstrates that cutting-edge competitive research can only be carried out efficiently within a research association", says Prof. Stoll. The German Research Foundation (SFB 642 and Research Unit 630) and the Research Department for Interfacial Systems Chemistry at RUB supported the work.
Further Information:
H. Bauke Albada, Florian Wieberneit, Ingrid Dijkgraaf, Jessica H. Harvey, Jennifer L. Whistler, Raphael Stoll, Nils Metzler-Nolte, Richard H. Fish:
The Chemoselective Reactions of Tyrosine-Containing G-Protein-Coupled Receptor Peptides with [Cp*Rh(H2O)3](OTf)2, Including 2D NMR Structures and the Biological Consequences.
In: Journal of the American Chemical Society; (25), pp 10321 - 10324, 07 June 2012, DOI 10.1021/ja303010k
Source: Ruhr-University, Bochum, RUB, Germany
Last update: 09.07.2012
Perma link: https://www.internetchemistry.com/news/2012/jul12/metal-peptide-complexes.php
More chemistry: index | chemicals | lab equipment | job vacancies | sitemap
Internetchemistry: home | about | contact | imprint | privacy
© 1996 - 2023 Internetchemistry
