To reduce fossil fuel consumption while simultaneously improving the carbon footprint of fuels and chemical products, the use of carbon dioxide as a carbon source could be an attractive option.
In the journal Angewandte Chemie, German researchers have now introduced a method by which carbon dioxide can be catalytically hydrogenated to make formic acid.
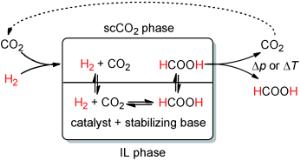
In this process, carbon dioxide is not only a starting material; it also acts - in a supercritical state - as the solvent for separation of the product.
This integrated approach makes it possible to directly obtain free formic acid as the product in a single step for the first time.
The hydrogenation of CO2 to formic acid (HCOOH) is a subject of intensive research because it offers direct access to chemical products based on waste products from the use of fossil fuels for energy. Formic acid is an important product in the chemical industry and has many applications, including agriculture, food technology, and the leather industry. It is also being contemplated as a potential hydrogen-storage material: vehicles powered by fuel cells could fill up with formic acid, from which the hydrogen could then be produced catalytically.
Homogeneous catalysts for the production of formic acid from CO2 have been investigated since the mid 1970s. The trouble with this process is that it involves an equilibrium reaction for which the equilibrium heavily favors the reactants. In order to suppress the constantly occurring back-reaction, the formic acid must be removed - in the form of a salt, adduct, or derivative - to take it out of the equilibrium. To obtain the desired free formic acid in the end, additional separation steps are thus required to separate the adducts from the catalyst and finally to release and isolate the formic acid.
A team led by Walter Leitner at the RWTH Aachen University has now developed a new concept that can be used to produce pure formic acid in a continuous process. The reaction and separation steps are integrated in a single processing unit.
Their trick is to use a two-phase reaction system that employs supercritical CO2 as the mobile phase and a liquid salt - an ionic liquid - as the stationary phase. The catalyst and the base used to stabilize the formic acid are both dissolved in the ionic liquid, which holds them both in the reactor. The CO2 flows through the reactor at pressures and temperatures above the critical values (74 bar, 31 C) and selectively removes the formic acid from the mixture. The dual role played by CO2 as both reactant and extractive phase has significant advantages: The product is continuously extracted and flushed from the reactor, which causes the equilibrium to readjust constantly. Once out of the reactor, the free formic acid can be obtained with high purity by decompression or washing. Ionic liquids do not dissolve in supercritical CO2, nor do the catalyst and base, so these do not contaminate the product. The process can run continuously. In laboratory experiments, stable operation was demonstrated for over 200 hours.
"Our results with formic acid demonstrate that the systematic implementation of modern solvent techniques in continuous reactor equipment makes it possible to perform conversions that cannot be achieved under conventional conditions," says Leitner. "Naturally we can't defeat thermodynamics in this way - but there are many possibilities for the integration of reactions and materials separation that may open new routes for more efficient and sustainable processes."
About the Author
Walter Leitner holds the Chair for Chemical Engineering and Petrochemistry at the Institut für Technische und Makromolekulare Chemie of RWTH Aachen University.Catalytic processes using CO2 have constituted a continuing theme in his research interests for over 20 years. He is an External Scientific Member of the Max Planck Institute for Coal Research, Mülheim an der Ruhr, and Scientific Director of CAT, the joint catalysis research center of Bayer and RWTH Aachen University.
Further Information:
Sebastian Wesselbaum, Dr. Ulrich Hintermair, Prof. Dr. Walter Leitner:
Continuous-Flow Hydrogenation of Carbon Dioxide to Pure Formic Acid using an Integrated scCO2 Process with Immobilized Catalyst and Base.
In: Angewandte Chemie International Edition; article first published online: 13 July 2012, DOI 10.1002/anie.201203185
Source: Angewandte Chemie International Edition, press release 30/2012
Last update: 06.08.2012
Perma link: https://www.internetchemistry.com/news/2012/aug12/formic-acid-from-co2.php
More chemistry: index | chemicals | lab equipment | job vacancies | sitemap
Internetchemistry: home | about | contact | imprint | privacy
© 1996 - 2023 Internetchemistry
