|
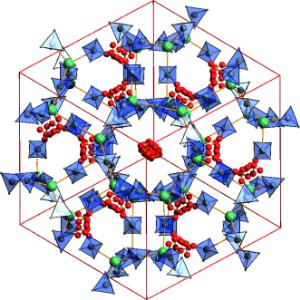
Highly occupied: A highly porous form of Mg(BH4)2 (see picture; Mg green, BH4 blue, unit cells shown in red) reversibly absorbs H2, N2, and CH2Cl2. At high pressures, this material transforms into an interpenetrated framework that has 79?% higher density than the other polymorphs. Mg(BH4)2 can act as a coordination polymer that has many similarities to metal–organic frameworks.
[Credit: Angewandte Chemie International Edition, Wiley-VCH] |
The perfect hydrogen storage material must store hydrogen efficiently and securely in a small volume, and should release it on demand. It must be rapidly refillable under mild conditions, while being as light and inexpensive as possible. One approach to this is solid-state storage. In such systems, hydrogen can be chemically bound, as in borohydride compounds, or it can be adsorbed as a molecule into a nanoporous material, as in some metal–organic frameworks.
The researchers have now found a material that can do both. It is a new, highly porous form of magnesium borohydride - the first light-metal hydride that is porous like a metal–organic framework and is capable of storing molecular hydrogen.
Magnesium borohydride (Mg(BH4)2) is one of the most promising materials for chemical hydrogen storage because it releases hydrogen at relatively low temperatures and can hold a high proportion by weight (about 15 %) of hydrogen. Two forms of this compound, a and ß, were previously known. The researchers have now made a third form, designated the ? form. Its pore volume comprises about 33 % of the structure, and its channels are wide enough to take up and store small gas molecules, such as nitrogen, dichloromethane, and most importantly hydrogen.
Interestingly, under high pressure this material converts into a nested, non-porous framework with a density that is nearly 80 % higher. This makes the d form the second densest in hydrogen content and more than twice as dense as liquid hydrogen. Furthermore, this conversion results in a 44 % reduction in volume, which is the largest contraction yet observed for a hydride.
“A combination of the chemical (through covalent bonding) and physical (through adsorption in the pores) storage of hydrogen seems to be difficult in practical applications,” explains Filinchuk. “However, this research has a broader impact, as it reveals a new class of hydride-based porous solids for storage and separation of various gases.”



