In an interesting bit of scientific serendipity, researchers at North Carolina State University have found that a chemical compound useful for studying the origins of intestinal birth defects may also inhibit the growth and spread of cancerous tumors.
During the screening of chemical compounds created by NC State chemist Dr. Alex Deiters, developmental biologist Dr. Nanette Nascone-Yoder found one of particular interest to her research: a compound that induced heterotaxia, a disordering or mirror-image "flipping" of internal organs, in the frog embryos she was studying.
Nascone-Yoder is particularly interested in the genetic processes involved in proper formation of the gut tube, which later becomes the intestinal tract.
"For the intestinal tract to form properly, it has to develop asymmetrically. This compound disrupts asymmetry, so it could be quite useful in helping us to determine when and where intestinal development goes wrong in embryos," Nascone-Yoder says.
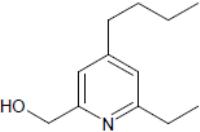
But the compound, dubbed "heterotaxin" by the researchers, had effects beyond just inducing heterotaxia.
"We also noticed that the compound prevents normal blood-vessel formation and prevents cells from migrating by increasing cellular adhesion - basically, the cells are stuck together and can't move."
Nascone-Yoder and her collaborators searched for known genetic pathways that could regulate all of these different events, and found that the pathway most likely to be affected by heterotaxin was the TGF-beta pathway. TGF-beta is known to play a role in the progression of cancerous tumors from normal to metastatic.
"This was exciting, because tumors have to have cells that can migrate and form a blood supply in order for the cancer to spread," Nascone-Yoder adds. "Heterotaxin inhibits those processes, which may make it a good 'lead' candidate for the development of an anti-tumor drug."
Indeed, collaborative experiments with NC State veterinary oncologist Marlene Hauck and cell biologist Philip Sannes showed that heterotaxin quenches the growth of canine tumor cells, and inhibits some of the changes required for human tumor cells to become migratory and invasive - at least in a petri dish. There is still work to do, but heterotaxin and future synthetic analogs could be the harbinger of a new class of cancer-fighting compounds.
The research was published in the Feb. 24, 2011 issue of Chemistry & Biology and was funded by the National Institutes of Health. Nascone-Yoder teaches in the Department of Molecular Biomedical Sciences, part of the College of Veterinary Medicine. The Department of Chemistry is part of the College of Physical and Mathematical Sciences.
Abstract
Disruptions of anatomical left-right asymmetry result in life-threatening heterotaxic birth defects in vital organs. We performed a small molecule screen for leftright asymmetry phenotypes in Xenopus embryos and discovered a novel pyridine analog, heterotaxin, which disrupts both cardiovascular and digestive organ laterality and inhibits TGF-ß-dependent left-right asymmetric gene expression. Heterotaxin analogs also perturb vascular development, melanogenesis, cell migration and adhesion, and indirectly inhibit the phosphorylation of an intracellular mediator of TGF-ß signaling. This combined phenotypic profile identifies these compounds as a novel class of TGF-ß signaling inhibitors. Notably, heterotaxin analogs also possess highly desirable anti-tumor properties, inhibiting epithelial-mesenchymal transition, angiogenesis and tumor cell proliferation in mammalian systems. Our results suggest that assessing multiple organ, tissue, cellular and molecular parameters in a whole organism context is a valuable strategy for identifying the mechanism of action of novel compounds.
Further Information:
Michael K. Dush, Andrew L. McIver, Meredith A. Parr, Douglas D. Young, Julie Fisher, Donna R. Newman, Philip L. Sannes, Marlene L. Hauck, Alexander Deiters, Nanette Nascone-Yoder:
Heterotaxin: A TGF-beta Signaling Inhibitor Identified in a Multi-Phenotype Profiling Screen in Xenopus Embryos.
In: Chemistry & Biology; Volume 18, Issue 2, 252-263, 25 February 2011, DOI 10.1016/j.chembiol.2010.12.008
Source: North Carolina State University, USA
Last update: 01.03.2011.
Perma link: https://www.internetchemistry.com/news/2011/mar11/heterotaxin.php
© 1996 - 2023 Internetchemistry