 |
Cobalt(III)-salen complexes with ammonium arm(s) have been used to catalyze the title transformation. Higher thermal decomposition temperatures than those of typical poly(propylene carbonate)s (PPCs) were observed for the stereogradient and the stereoblock PPCs obtained by this method.
[Credit: Angewandte Chemie] |
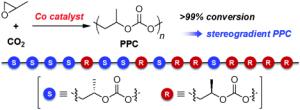
Poly(propylene carbonate) is used as a binding agent and as a component of biodegradable plastics. It is made from propylene oxide and carbon dioxide in a catalytic process. Propylene oxide contains three carbon atoms, two of which form a ring together with an oxygen atom. This ring opens during polymerization. Propylene oxide exists in two forms that are mirror images of each other; these are designated as the S and R stereoisomers.
Poly(propylene carbonate)s that are made primarily of one of the two forms or have both forms in an alternating pattern have been made before. Nozaki’s group has now been the first to synthesize both a stereoblock and a stereogradient. A stereoblock copolymer is a chain, half of which is made of the S form and the other half of the R form. In a stereogradient copolymer, the composition changes gradually from the S form to the R form.
Making a block copolymer is theoretically relatively easy because use of an asymmetric catalyst causes one of the two forms of building block to be used preferentially, so it is built into the polymer chains first; the less favorable form is incorporated afterward. In the case of poly(propylene carbonate), however, this process isn’t so trivial because once the favored form of the propylene oxide is converted to a polymer, the other form decomposes instead of polymerizing. The Japanese scientists found a special asymmetrical cobalt complex that allows nearly complete conversion to the polymer. Although the catalyst prefers the S form, it also ensures that it is more favorable for the R form to polymerize than to decompose.
The researchers experimented further with variations on the cobalt complex. A special ammonium side branch on a ligand brought success: It balances the degree of preference of the catalyst for the S form over the R form so that the R form begins to be incorporated into the polymer chain as the amount of the S form decreases. This allows the formation of the stereogradient copolymer. Interestingly, both of the new types of poly(propylene carbonate), stereoblock and stereogradient, are significantly more heat-tolerant than pure S or R polymers or mixtures of the two.



