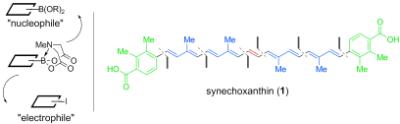
|
The first total synthesis of the antioxidant carotenoid synechoxanthin was achieved through a novel iterative cross-coupling approach in which the polarity of the bifunctional building blocks is reversed to match the preferred polarity for CC bond formation (see scheme). The convergent, stereocontrolled, and flexible nature of this synthesis enables systematic studies of the biological activities of this natural product.
[Credit: Angewandte Chemie International Edition, DOI: 10.1002/anie.201102688] |
"There's pre-installed functionality and stereochemistry, so everything is set in the building blocks, and all you have to do is couple them together," said graduate student Seiko Fujii, the first author of the paper.
However, ICC has been limited to only molecules with one type of polarity. Now, the group has developed reverse-polarity ICC, which allows a chemist to optimize the ICC process to match the target molecules' electronic structure. The reversal in polarity enables a whole new class of building blocks, so researchers can synthesize molecules more efficiently and even construct molecules that standard ICC cannot.
For example, in the paper, the group used the new method to make synechoxanthin (pronounced sin-ecko-ZAN-thin), a molecule first isolated from bacteria in 2008 that shows great promise as an antioxidant. Studies suggest that synechoxanthin allows the bacteria that produce it to live and thrive in highly oxidative environments.
"We as humans experience a lot of oxidative stress, and it can be really deleterious to human health," said Burke, who also is affiliated with the Howard Hughes Medical Institute. "It can lead to diseases like cancer and atherosclerosis and neurodegenerative disorders. Evidence strongly suggests that synechoxanthin is a major part of the bacteria's solution to this problem. We're excited to ask the question, what can we learn from the bug? Can it also protect a human cell?"
Studies on the activity of synechoxanthin have been limited by the difficulty of extracting the molecule from bacterial cultures. Burke's group successfully synthesized it from a mere three types of readily available, highly stable, non-toxic building blocks. Thanks to the ease of ICC, they can produce relatively large quantities of synechoxanthin for study as well as derivatives to test against the natural product.
"Because this building-block-based design is inherently flexible, once we've made the natural product, we can make any derivative we want simply by swapping in one different building block, and then using the reverse-polarity ICC to snap them together," Burke said. "That's where synthesis is so powerful. Oftentimes, the cleanest experiment will require a molecule that doesn't exist, unless you can piece it together."
Researchers can also use blocks that have been "tagged" with a fluorescent or radioactive dye to make it easier to study the molecule and its activity. For example, Fujii next plans to synthesize both synechoxanthin and its apolar derivative with tags so that NMR imaging can reveal its location and orientation within a cell's membrane, possibly providing clues to its activity.
"After we have all these molecules in hand, we're really excited to test the antioxidant activity of them in a model membrane," Fujii said.
The National Institutes of Health and the Howard Hughes Medical Institute supported this work.



