New Haven, Conn. - Yale University scientists have streamlined the process for synthesizing a family of compounds with the potential to kill cancer and other diseased cells, and have found that they represent a unique category of anti-cancer agents. Their discovery appearsed in the online edition of the Journal of the American Chemical Society.
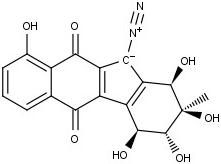
The team studied a family of compounds known as the kinamycins, which are naturally produced by bacteria during metabolism and are known for their potent toxicity. For years scientists have guessed that a core structure common to the different compounds within the group was responsible for this toxicity. Until now, chemists could not study the core structure because there was no simple way to create it in the laboratory.
Now the Yale team has developed a new method to recreate this structure that allows them to synthesize the kinamycins with much greater efficiency than previously possible. While scientists have produced kinamycins in the laboratory in the past, the Yale team was able to halve the number of steps required to go from simple, easily obtainable precursors to the complete molecule - from 24 down to 12.
"By shortening the synthesis we can now prepare these molecules in the quantities required for further studies, including animal studies and even clinical trials," said Seth Herzon, assistant professor of chemistry and lead author of the study.
Working with researchers at the Yale School of Medicine and the Yale Chemical Genomics Screening Facility, the team has begun testing several of the compounds against cancer cells, with promising preliminary results. Next, they will work to understand the exact mechanism that makes the compounds - which are benign on their own - highly toxic once they penetrate cells.
"The key to success will be whether we can develop selectivity - whether we can kill cancer cells in the presence of non-cancerous tissue," Herzon said. "Based on what we already know about the chemical reactivity of these molecules, I"m optimistic we can do this."
The reactive core of the kinamycins also plays a key role in another compound the team is studying, called lomaiviticin A, which is even more toxic and could prove even more effective in destroying cancer cells. "Lomaiviticin A is the big fish. It"s more potent than the kinamycins, but it"s also much harder to synthesize," Herzon said.
Both the kinamycins and lomaiviticin A are unique in their toxicity profiles, Herzon said, representing a new category of anti-cancer agents.
"There"s no close analogy to draw from to predict how these molecules will behave, which will make it especially interesting to see where this research takes us," Herzon said. "This research involves a lot of exciting chemistry, but it also has real applications in biology and human medicine."
Other authors of the study include Christina Woo, Liang Lu, Shivajirao Gholap and Devin Smith, all of Yale University.
Funding for this research was provided by Yale University and Eli Lilly.
Further Information:
Christina M. Woo, Liang Lu, Shivajirao L. Gholap, Devin R. Smith and Seth B. Herzon:
Development of a Convergent Entry to the Diazofluorene Antitumor Antibiotics: Enantioselective Synthesis of Kinamycin F.
In: Journal of the American Chemical Society; Publication Date (Web): February 8, 2010, DOI 10.1021/ja910769j
Source: Yale University, USA
Last update: 15.02.2010
Perma link: https://www.internetchemistry.com/news/2010/feb10/kinamycin-f.php
More chemistry: index | chemicals | lab equipment | job vacancies | sitemap
Internetchemistry: home | about | contact | imprint | privacy
© 1996 - 2023 Internetchemistry
