|
Chemistry News Archive May 2011
|
Chemistry News May 2011
News of the year 2011 in the fields of chemistry and chemistry-related topics like biochemistry, nantechnology, medicinal chemistry etc.
Main focus: press releases, scientific research results and summaries of chemistry articles, that are published in chemistry journals.
Please send us a eMail to publish your press release here!
|
|
| | | Chemistry: |  |
|
Researchers have expanded the range of monomer pairs used in their established metal-free click polymerization of aroylacetylene-azides to propiolate-azides.
|
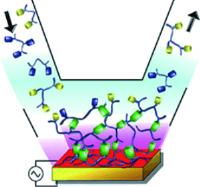 |
Signpost for chemical snaps: copper ions as morphogens for the formation of polymer films by click chemistry. Image: A polymer film is obtained by the Cu(I)-catalyzed Sharpless click reaction between two polymers, bearing either azide or alkyne groups, both present simultaneously in a Cu(II) solution (see picture). The Cu(I) morphogen is generated at an electrode by applying an adequate potential. This concept can be extended to supramolecular films formed by coordination complexes [Credit: Angewandte Chemie International Edition].
|
|
The American Physical Society has released a new assessment - Direct Air Capture of CO2 with Chemicals - to better inform the scientific community on the technical aspects of removing carbon dioxide from the atmosphere.
|
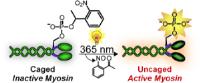 |
Nano-motor with a light switch: light-triggered myosin allows real-time study of cells. [Credit: Angewandte Chemie International Edition]
|
|
Olfactory properties and gas-phase structures of Cassyrane stereoisomers.
|
| | | Physics: |  |
 |
CIC nanoGUNE develops Nano-FTIR-nanoscale infrared spectroscopy with a thermal source. Image: The tip is illuminated with the broadband infrared radiation from of a thermal source and the backscattered light is analyzed with a Fourier spectrometer, yielding local infrared spectra with a spatial resolution better than 100 nm. The displayed graph shows infrared spectra of differently processed oxides in an industrial semiconductor device [Copyright F. Huth, CIC nanoGUNE].
|
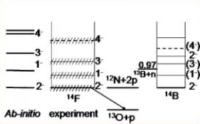 |
A recent discovery of an extremely exotic, short-lived nucleus called fluorine-14 in laboratory experiments may indicate that scientists are gaining a better grasp of the rules of strong interaction. Image: This graph shows the flourine-14 supercomputer predictions (far-left) and experimental results (center) [Credit: James Vary].
|
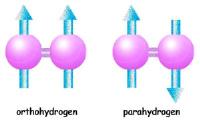 |
Nuclear magnetic resonance with no magnets. Image: Hydrogen molecules consist of two hydrogen atoms that share their electrons in a covalent bond. In an orthohydrogen molecule, both nuclei are spin up. In parahydrogen, one is spin up and the other spin down. The orthohydrogen molecule as a whole has spin one, but the parahydrogen molecule has spin zero [Credit: Lawrence Berkeley National Laboratory].
|
| | | Chemistry and Nanotechnology: |  |
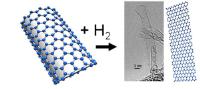 |
An international research team has discovered a new method to produce belts of graphene called nanoribbons. By using hydrogen, they have managed to unzip single-walled carbon nanotubes. The method also opens the road for producing nanoribbons of graphane, a modified and promising version of graphene. Image: Reaction of single-walled carbon nanotubes (SWNTs) with hydrogen gas was studied in a temperature interval of 400–550 °C and at hydrogen pressure of 50 bar [Credit: The Swedish Research Council].
|
 |
Berkeley Lab researchers use nanoantenna to enhance plasmonic sensing. Image: This is a scanning electron microscopy image showing a palladium nanoparticle with a gold antenna to enhance plasmonic sensing [Image courtesy of Alivisatos group].
|
| | | Chemistry and Environment: |  |
|
New evidence for natural synthesis of silver nanoparticles.
|
| | | Chemistry and Research: |  |
 |
Which is more efficient at harvesting the sun's energy, plants or solar cells? This salient question and an answer are the subject of an article published in the May 13, 2011 issue of the journal Science. Image: Photosynthesis or photovoltaics: Weighing the impact [Credit: Bob Blankenship].
|
| | | More News (open access): |
Following your steak’s history from pasture to plate
|

|
|
Steaks sold as “grass-fed” or “grass-finished”
can be checked for authenticity using a test that reconstructs the
dietary history of cattle. [Credit: iStock] |
The package on a supermarket steak may say
“grass-fed” or “grass-finished,” but how can a consumer know whether
the cow spent its days grazing peacefully on meadow grass or actually
gorged on feedlot corn? In ACS’s Journal of Agricultural and Food
Chemistry, scientists are now reporting the development of a method
that can reconstruct the dietary history of cattle and authenticate
the origins of beef. Frank J. Monahan and colleagues
note that consumers are increasingly concerned about the origins and
labeling of meat, as they seek assurance about the meat’s safety or
prepare to pay premium prices for specialty meats that are raised
locally or certified as organic. “An example of such a product is
pasture-fed beef,” they write, “often marketed as superior
nutritionally as a result of increased levels of omega-3 fatty
acids...arising from the consumption of grass.” To
reconstruct the diet of cattle, the researchers analyzed the
proportions of different types of oxygen, nitrogen, hydrogen, and
sulfur in the animals’ muscle tissue and tail hair. Specific diets
(for instance, a diet that switched from mostly grass to corn at the
end of the cow’s life) leave a distinctive “fingerprint” of these
elements in cattle tissue. The fingerprint in muscle represents the
animal’s overall lifetime diet, while quicker-growing tissue in tail
hair can reveal more recent dietary changes. Monahan and colleagues
say the fingerprints “provide a powerful tool to reconstruct changes
in feed components offered to animals over periods of over a year and
thus a tool to verify farm production practices.”
The authors acknowledge funding from the Irish Department of
Agriculture, Fisheries and Food. Journal of
Agricultural and Food Chemistry: "Beef
Authentication and Retrospective Dietary Verification Using Stable
Isotope Ratio Analysis of Bovine Muscle and Tail Hair" [J. Agric.
Food Chem., 2011, 59 (7), pp 3295–3305; DOI: 10.1021/jf1040959]. |
“Liquid smoke” from rice shows potential health benefits
Liquid smoke flavoring made from hickory
and other wood - a mainstay flavoring and anti-bacterial
agent for the prepared food industry and home kitchens - may
get a competitor that seems to be packed with antioxidant,
antiallergenic and anti-inflammatory substances, according
to a new study in ACS’ Journal of Agricultural and Food
Chemistry. It is the first analysis of liquid smoke produced
from rice hulls, the hard, inedible coverings of rice
grains. Mendel Friedman, Seok Hyun Nam and
colleagues explain that wood from trees is typically used to
produce liquid smoke, added to meat and other foods for a
smoky taste. But other types of plants can also be burned to
make the popular seasoning. Rice is a prime candidate, with
680 millions tons produced worldwide each year. Hulls
account for 136 million tons of that amount and often go to
waste. The researchers wondered rice hulls could be put to
good use in a liquid form as a food flavoring, and did the
first studies needed to determine if rice hull smoke is safe
enough for food use. The scientists found
that liquid smoke from rice hulls may be healthful. Their
tests on laboratory cell cultures found that liquid rice
hull smoke worked as an antioxidant that could help fight
off diseases. It also helped prevent inflammation, which is
associated with many different health problems did not
trigger an allergic response. “New food uses of a major
agricultural byproduct may benefit the environment, farmers,
and consumers,” the report stated. “However, it is necessary
to demonstrate that rice hull smoke is safe. The present
study was designed to contribute to this assessment.”
The authors acknowledge funding from the Rural Development
Administration, Republic of Korea.
Journal of Agricultural and Food Chemistry: "Composition
of Liquid Rice Hull Smoke and Anti-Inflammatory Effects in
Mice" [J. Agric. Food Chem., 2011, 59 (9), pp 4570–4581;
DOI: 10.1021/jf2003392]. |
Toward a vaccine for methamphetamine abuse
|

|
|
A new vaccine now in development may help treat
addiction to methamphetamine. [Credit: U.S. Drug
Enforcement Administration (DEA)] |
Scientists are reporting development of three
promising formulations that could be used in a vaccine to treat
methamphetamine addiction - one of the most
serious drug abuse problems in the U.S. The report appears in the
Journal of the American Chemical Society.
In the paper, Kim Janda and colleagues note that
methamphetamine use and addiction cost the U.S. more than $23 billion
annually due to medical and law enforcement expenses, as well as lost
productivity. The drug, also called “meth” or “crystal meth,” can
cause a variety of problems including cardiovascular damage and death.
Meth is highly addictive, and users in conventional behavioral
treatment programs often relapse. Previously tested meth vaccines
either are not effective or are very expensive. To overcome these
challenges, the researchers made and tested new vaccine formulations
that could potentially be effective for long periods, which would
drive down costs and help prevent relapse.
The group found that three of the new formulations
that produced a good immune response in mice (stand-ins for humans in
the lab) were particularly promising. “These findings represent a
unique approach to the design of new vaccines against methamphetamine
abuse,” say the researchers.
The authors acknowledge funding from the National
Institute on Drug Abuse and The Skaggs Institute for Chemical Biology.
Journal of the American Chemical Society: "Impact
of Distinct Chemical Structures for the Development of a
Methamphetamine Vaccine" [J. Am. Chem. Soc., 2011, 133 (17), pp
6587–6595; DOI: 10.1021/ja108807j]. |
The “$1,000 genome” may cost $100,000 to understand
Advances in technology have almost lifted
the curtain on the long-awaited era of the “$1,000 genome” -
a time when all the genes that make up a person can be
deciphered for about that amount - compared to nearly $1
million a few years ago. But an article in the current
edition of Chemical & Engineering News (C&EN), ACS’ weekly
newsmagazine, raises the disconcerting prospect that a price
tag of $100,000, by one conservative estimate, is necessary
to analyze that genetic data so it can be used in
personalized medicine – custom designing treatments that fit
the patient’s genetic endowment. In the
article, C&EN Senior Editor Rick Mullin explains that while
the cost of sequencing genes has dropped dramatically, the
cost of analyzing genomic data so that it can be put to
practical use in medicine has hardly budged. Today,
assessing the genetic predispositions to disease means
costly data analysis by specialists from several research
areas, including molecular and computational biology,
genetics, pathology and clinical science.
Mullin, however, cites several trends in bioinformatics that
are opening the door to collection and processing of genetic
data more economically and efficiently. One trend is to
incorporate genomic analysis in commercial drug discovery
and development efforts from the beginning. Another way to
ease the burden is to reduce the amount of data that is
generated - one instrument company recently developed a
brand-new sequencing technology that generates much smaller
data files, for example. Pharma researchers also are
collaborating and sharing data like never before, and some
of them are making use of public cloud computing and free,
open-source software. Chemical &
Engineering News: "The
Next Generation in Genome Sequencing" [May 9, 2011]. |
New evidence that caffeine is a healthful antioxidant in coffee
|

|
|
The caffeine found in coffee,
tea, and other foods appears to provide healthful benefits by
acting as a powerful antioxidant.
Credit: iStock |
Scientists are reporting an in-depth analysis of
how the caffeine in coffee, tea, and other foods seems to protect
against conditions such as Alzheimer’s disease and heart disease on
the most fundamental levels. The report, which describes the chemistry
behind caffeine’s antioxidant effects, appears in ACS’ The Journal of
Physical Chemistry B.
Annia Galano and Jorge Rafael León-Carmona describe
evidence suggesting that coffee is one of the richest sources of
healthful antioxidants in the average person’s diet. Some of the
newest research points to caffeine (also present in tea, cocoa, and
other foods) as the source of powerful antioxidant effects that may
help protect people from Alzheimer’s and other diseases. However,
scientists know little about exactly how caffeine works in scavenging
the so-called free radicals that have damaging effects in the body.
And those few studies sometimes have reached contradictory
conclusions.
In an effort to bolster scientific knowledge about
caffeine, they present detailed theoretical calculations on caffeine’s
interactions with free radicals. Their theoretical conclusions show
“excellent” consistency with the results that other scientists have
report from animal and other experiments, bolstering the likelihood
that caffeine is, indeed, a source of healthful antioxidant activity
in coffee.
The Journal of Physical Chemistry B:
"Is
Caffeine a Good Scavenger of Oxygenated Free Radicals?" [J. Phys.
Chem. B, 2011, 115 (15), pp 4538–4546; DOI: 10.1021/jp201383y]. |
Natural protection against radiation
|

|
|
A substance similar to resveratrol — an
antioxidant found in red wine, grapes, and nuts — could protect
against radiation sickness.
Credit: iStock |
In the midst of ongoing concerns about radiation
exposure from the Fukushima nuclear power plant in Japan, scientists
are reporting that a substance similar to resveratrol — an antioxidant
found in red wine, grapes and nuts — could protect against radiation
sickness. The report appears in ACS Medicinal Chemistry Letters.
Michael Epperly, Kazunori Koide and colleagues
explain that radiation exposure, either from accidents (like recent
events in Japan) or from radiation therapy for cancer, can make people
sick. High doses can even cause death. The U.S. Food and Drug
Administration is currently evaluating a drug for its ability to
protect against radiation sickness, but it is difficult to make in
large amounts, and the drug has side-effects that prevent its use for
cancer patients. To overcome these disadvantages, the researchers
studied whether resveratrol — a natural and healthful antioxidant
found in many foods — could protect against radiation injuries.
They found that resveratrol protected cells in
flasks but did not protect mice (stand-ins for humans in the
laboratory) from radiation damage. However, the similar natural
product called acetyl resveratrol did protect the irradiated mice. It
also can be produced easily in large quantities and given orally. The
authors caution that it has not yet been determined whether acetyl
resveratrol is effective when orally administered.
ACS Medicinal Chemistry Letters: "The
Use of 3,5,4′-Tri-O-acetylresveratrol as a Potential Prodrug for
Resveratrol Protects Mice from γ-Irradiation-Induced Death" [ACS
Med. Chem. Lett., 2011, 2 (4), pp 270–274; DOI: 10.1021/ml100159p]. |
Cola detectives test natural flavoring claims for pricey soft drinks
|

|
|
Pricey cola drinks that claim to be made from
natural kola nuts can be checked for authenticity using a new
test.
Credit: iStock |
Scientists are reporting development and successful
testing of a new way to determine whether cola drinks — advertised as
being made with natural ingredients and sold at premium prices —
really do contain natural flavoring. The report appears in ACS’
Journal of Proteome Research.
In the study, Pier Giorgio Righetti and colleagues
explain that cola drinks purportedly made from natural cola nuts are
becoming popular and are sold in many natural food stores. Genuine
cola “nuts” are seeds from the fruit of the cola tree, which is native
to African rainforests, and they are expensive to harvest and ship. In
West African cultures, people include the nuts in ceremonies and offer
them to guests. The nuts also have possible health benefits — they may
help treat whooping cough, asthma, migraines and dysentery. Most soft
drink manufacturers don’t use cola nuts today, but a select few are
starting to advertise cola as a natural ingredient in their products —
and charge extra for it. To see whether consumers are getting what
they pay for, the scientists set out to find a way to finger the
drinks with real natural extracts.
The group found that testing for proteins was an
accurate way to verify natural flavoring claims. They detected plant
proteins in a drink claiming to have “organic agave syrup and cola nut
extracts”. On the other hand, Coca Cola products — which do not claim
to include cola extract — have no protein. The scientists say, “The
identifications here obtained represent the quality mark of this
beverage and, in a way, give a certificate of authenticity.”
Journal of Proteome Research: "Going
nuts for nuts? The trace proteome of a Cola drink, as detected via
combinatorial peptide ligand libraries" [J. Proteome Res., Article
ASAP, 2011; DOI: 10.1021/pr2001447]. |
New woes for silicones in cosmetics and personal care products
At a time when cosmetics, shampoos, skin
creams, and other personal care products already are going
green / with manufacturers
switching to plant-derived extracts and other natural
ingredients government regulators in Canada are adding
to the woes of the silicone-based ingredients long used in
these products. That’s the topic of an article in the
current edition of Chemical & Engineering News (C&EN), ACS’
weekly newsmagazine.
C&EN Senior Correspondent Marc S. Reisch
points out that manufacturers have used silicones for
decades in an array of personal care products.
Antiperspirants and underarm deodorants account for about
half the entire U.S. personal care market for silicones.
Manufacturers voluntarily stopped using one type of silicone
ingredient in personal care products over the last decade.
Now government regulators in Canada are proposing
regulations limiting use of another widely used type of
silicone ingredient. They cite concerns that the ingredients
might built up in the environment and harm wildlife.
The article notes that some
manufacturers, despite the concerns, are sticking with the
traditional ingredients, termed cyclic methylsiloxanes.
Others are using the concerns as a basis for jumping on the
natural ingredient bandwagon and reformulating their
products with other silicones or as “silicone-free.”
Chemical & Engineering News: "Storm
Over Silicone" [Volume 89, Number 18, pp 10-13, DOI
10.1021/CEN042611142704]. |
New high-performance lithium-ion battery top candidate for electric cars
Scientists are reporting development of
an advanced lithium-ion battery that is ideal for powering
the electric vehicles now making their way into dealer
showrooms. The new battery can store large amounts of energy
in a small space and has a high rate capacity, meaning it
can provide current even in extreme temperatures. A report
on this innovation appears in ACS’ Journal of the American
Chemical Society.
Bruno Scrosati, Yang-Kook Sun, and
colleagues point out that consumers have a great desire for
electric vehicles, given the shortage and expense of
petroleum. But a typical hybrid car can only go short
distances on electricity alone, and they hold less charge in
very hot or very cold temperatures. With the government push
to have one million electric cars on U.S. roads by 2015, the
pressure to solve these problems is high. To make electric
vehicles a more realistic alternative to gas-powered
automobiles, the researchers realized that an improved
battery was needed.
The scientists developed a high-capacity,
nanostructured, tin-carbon anode, or positive electrode, and
a high-voltage, lithium-ion cathode, the negative electrode.
When the two parts are put together, the result is a
high-performance battery with a high energy density and rate
capacity. “On the basis of the performance demonstrated
here, this battery is a top candidate for powering
sustainable vehicles,” the researchers say.
The authors acknowledge funding from WCU
(World Class University) program through the Korea Science
and Engineering Foundation.
Journal of the American Chemical
Society: "An
Advanced Lithium Ion Battery Based on High Performance
Electrode Materials" [J. Am. Chem. Soc., 2011, 133 (9),
pp 3139–3143; DOI: 10.1021/ja110522x]. |
|
|



